News
Sino Biopharm Was Selected as Top 20 Biopharmaceutical Enterprises with Biggest Innovation Potential in 2022
Release time:2022-10-13
Recently, the "2022 Top 100 China Biopharmaceutical Enterprises List" which includes series of lists launched by Times Data of Times Media Group has been launched one after another, and on October 10, the " Top 20 Biopharmaceutical Enterprises with Biggest Innovation Potential in 2022 List" was released, and Sino Biopharm was ranked fourth.
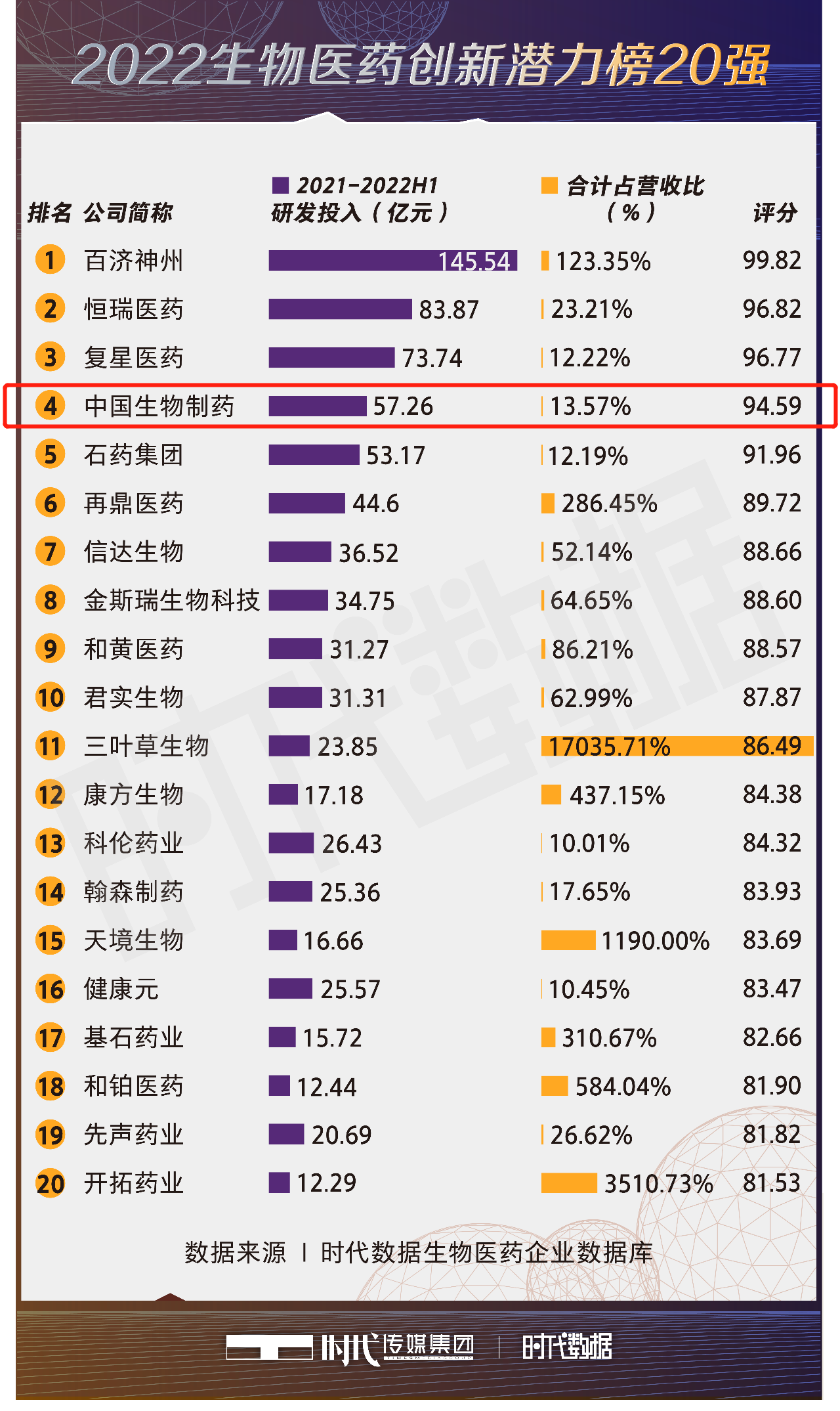
The list is based on the "Times Data Biopharmaceutical Enterprise Database", which analyzes the R&D investment and the ratio of R&D investment to revenue of major biopharmaceutical companies for the whole year of 2021 and the first half of 2022 through big data modeling and analysis, and provides a comprehensive score.
As an innovation-led biopharmaceutical company, Sino Biopharm has been increasing its R&D investment in innovation, and has invested more than RMB 10 billion in R&D in the past four years. In the first half of 2022, the Group invested RMB 2.19 billion in R&D, of which the R&D investment in innovative drugs accounted for 73.7%.
At the same time, Sino Biopharm's innovative R&D strength has been widely recognized both at home and abroad. So far this year, Sino Biopharm has been ranked second in the "China Big Pharma Innovation Power TOP10", the first tier of "Top 100 Chinese Pharmaceutical Innovation Enterprises 2022", the top of the "50 China Pharmaceutical Innovation and Upgrading Models", and the 40th place of "Top 50 Global Pharmaceutical Companies 2022", etc.
Heavyweight innovative products are expected to be launched
In recent years, Sino Biopharm has been broadening its research and development pipeline, forming a research and development pattern with liver disease drugs and anti-tumor drugs as the mainstay, as well as digestive system, anti-infection, respiratory system and other areas of synergistic development.
Since 2022, a number of Class 1 innovative drugs developed by the Group have been approved for clinical trials, such as TDI01, an oral small molecule drug with a new target and a new mechanism, which has been listed as a major new drug creation in the 13th Five-Year Plan of China, and in addition to the clinical trial for idiopathic pulmonary fibrosis previously approved in China, the clinical trials for pneumoconiosis and anti-neoplastic pneumonia were approved this year. In addition, a number of Class 1 innovative drugs such as FHND5071, TQB2930 and TQC2938 injection were also approved for clinical trials this year.
Currently, Sino Biopharm's pipeline is rich in four major areas: anti-tumor, surgical/analgesic, liver disease and respiratory, with three innovative drugs approved for marketing, two innovative drugs reported for production, more than 50 innovative products in various clinical stages and more than 60 innovative drug candidates in pre-clinical stage.
The new class 1 drugs under development involve PD-(L)1, ALK, EGFR, PI3K, CDK4/6, MEK, c-Met, DPP4, JAK and other popular targets, covering anti-tumor, liver disease, digestive system, anti-infection, respiratory system and other disease areas.
Among them, the potentially heavyweight Yilisheng/Yilisu® (Ebegastine α injection), which has filed marketing applications in China and the US respectively, is expected to be approved in China in the first half of 2023. In addition, there are 3 oncology innovative drugs, ALK, ROS1 and PI3K inhibitors, which will be marketed in the next 3 years.
Accelerating the introduction of multiple innovative and differentiated new drugs
While increasing its own R&D, Sino Biopharm is also introducing more innovative products through M&A investment and external introduction.
So far this year, Sino Biopharm has entered into collaborations with a number of domestic and international pharmaceutical companies, including Inventiva, Anyuan Pharmaceuticals and XtalPi, to develop or commercialize a number of innovative and differentiated products.
On September 21, the Group's subsidiary CTTQ signed a formal license agreement with Inventiva to develop, manufacture and commercialize lanifibranor for the treatment of nonalcoholic steatohepatitis (NASH) and other underlying metabolic diseases in China, Hong Kong, Macau and Taiwan (Greater China region).
Lanifibranor is a peroxisome proliferator-activated receptor (PPARs) allosteric agonist that improves insulin sensitivity, activates macrophages, and reduces liver fibrosis and inflammatory gene expression by activating three functional receptor subtypes in vivo: alpha, gamma and delta. The product is currently in Phase III clinical trials in the U.S. and is expected to be the first oral drug approved by the FDA for the treatment of NASH.
At present, Sino Biopharm has established four strategies: organizational integration, comprehensive innovation, internationalization and digitalization, of which the internationalization strategy consists of two major parts:"bringing in" and "going out". In terms of "bringing in", invoX, a wholly-owned European subsidiary, has been established to focus on differentiated dual-antibody, triple-antibody platform, mRNA/DNA and other technologies to carry out a comprehensive global strategic layout. The Group has also been investing in internal innovation and R&D to provide a new growth engine for the goal of becoming one of the "Top 30 Global Companies".